Crop Production

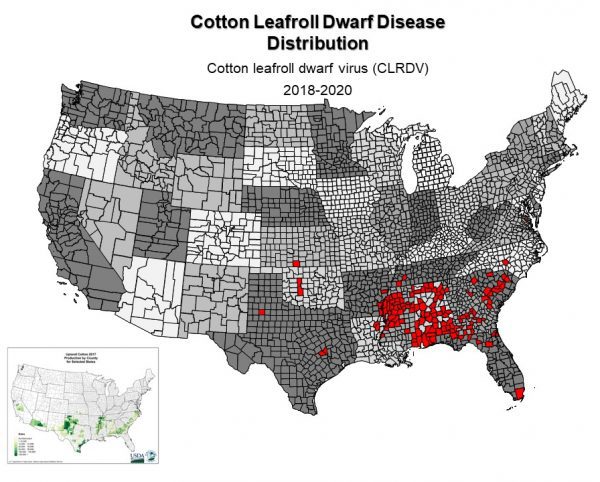
Figure 1. Known distribution of US-CLRDV across the cotton belt.
Cotton leafroll dwarf virus (US-CLRDV) (genus Polerovirus, family Luteoviridae) was first identified in the United States in samples collected from Barbour County, Alabama. Since then, the virus has spread across the cotton belt as far west as Texas and north to Virginia. Whole genome sequences were derived from symptomatic samples collected during 2018 from Barbour and Macon Counties, and their analysis showed that the strain of CLRDV found in the United States is different from the typical and atypical strains found in South America, identifying the CLRDV strain in Alabama as unique. Whole genome sequences of US-CLRDV isolates collected from other states have been completed and are similar across the cotton belt, suggesting a single introduction of the virus into the southeast United States. The disease caused by US-CLRDV has been named cotton leafroll dwarf disease (CLRDD).
Symptoms
Symptoms attributed to cotton blue disease (CBD) were first described in Africa around 1949. More recently, CBD has been identified in South America, Argentina, India, Thailand, and Timor- Leste, where the causal agent was identified as CLRDV, using PCR-based diagnostics. Cotton plants infected by the typical strain of this virus exhibit stunting due to internodal shortening, leaf rolling, petiole and vein reddening, distorted new growth, reduced flower and boll size, and sterility. In Brazil, during 2006, a new strain of the virus was seen in fields of CBD-resistant cotton. Virus-infected plants showed mild CBD symptoms associated with red, withered leaves and accentuated verticality. The new strain was identified as atypical CLRDV. Another outbreak was reported in 2009–2010 in Argentina where CBD-resistant cultivars showed severe disease symptoms. Worldwide, CLRDV is generally regarded as the second-most damaging virus disease to commercial cotton, second to cotton leaf curl virus, and yield losses of up to 80 percent in susceptible varieties have been reported from South America.
Symptoms observed early in the season (June–July) consist of stunted plants with distorted leaves and can easily be confused with other disorders, particularly thrips feeding injury (figure 2). Later in the season, symptoms become variable based on plant age at infection, variety, environmental conditions, and nutritional status. Symptoms tend to be more severe in June- planted than May-planted cotton and consist of rugosity (puckering) and distortion of the juvenile leaves in the main and lateral shoot terminals (figure 3), along with upward or downward cupping of leaves (figures 4 and 6), and red to maroon discoloration of leaf veins (figure 5). Aphid feeding alone can cause leaf curling, and reddish to maroon discoloration of petioles and stems just below the shoot terminals with shortened internodes in a zig-zag pattern can occur (figure 9). The maroon discoloration of petioles and stems may be accompanied by wilted foliage that is warmer than normal to the touch (figure 7). Severely damaged plants develop a triangular shape with elongated whips or shoots and many fruiting nodes and squares, which are later shed and result in few blooms and bolls set on the main terminal whips and lateral branches (termed accentuated verticality) (figures 10 and 11). Failure of diseased plants to retain squares and set blooms or bolls on the whips can cause top crop loss. Usually when this occurs, there are clusters of symptomatic plants; occasionally an entire field will display these symptoms. Shedding of nearly mature bolls has also been observed as have severely stunted, nonboll-bearing plants.
Overall, symptoms on mature plants infected around cutout with open bolls throughout the canopy are often limited to leaf rugosity and distortion of leaves in the shoot terminals and regrowth at the plant base or a bronze coloration of the foliage and folding of the leaves (figure 8). In contrast, a noticeable demarcation between a normal boll set in the lower to midcanopy and the production of the elongated, sterile terminal whips or accentuated verticality (figure 10) on later-planted cotton infected during flowering and early boll set indicate that significant yield loss occurred.
- Figure 2. Stunted plant due to early season infection. Plants in figures 2–8 tested positive for CLRDV.
- Figure 3. Leaf rugosity or puckering along with yellow veins in shoot terminal. (Photo credit: D. Schrimsher)
- Figure 4. Upward cupping of juvenile leaves.
- Figure 5. Red to maroon leaf midveins.
- Figure 6. Rugosity and downward leaf roll on adventitious shoots at base of mature cotton.
- Figure 7. Maroon petioles and stems with wilted foliage.
- Figure 8. Bronze coloration and folding of foliage.
- Figure 9. Advanced symptoms of CLRDV-related virus disease in Alabama cotton include red to maroon discoloration of leaf petioles and terminal shoots with shortened internodes and sometimes a pronounced zig-zag growth pattern.
- Figure 10. Advanced symptoms also include elongated, often bare, terminal whips or shoots with normal boll set in lower and midcanopy.
- Figure 11. CLRDV-infected cotton exhibiting the triangular or pyramidal shape with elongated terminal shoots (accentuated verticality) with a dense leaf canopy. Notice the absence of blooms and open bolls in the upper canopy.
Vector
As with other Polerovirus species, CLRDV is transmitted by the cotton aphid (Aphis gossypii) in a persistent and circulative manner. Alates can transmit the virus in as little as 40 seconds, and viruliferous aphids can transmit for up to 12 days. Intensive insecticide programs directed at controlling this insect pest do not reduce the virus spread to fields when aphids colonize the crop because insecticides will not kill the aphid vector before they can transmit the virus. An insect disease caused by the fungus Neozygites fresenii kills most cotton aphids in early summer; however, aphid populations persisting into late October have been seen recently in shoot terminals of Alabama cotton. When cotton aphid populations exceed the established treatment threshold, recommended insecticides should be applied as needed.
Impact
The impact of CLRDD on lint yield in Alabama was assessed in 2019 and 2020 at Alabama Agricultural Experiment Stations (AAES) in southwest, central, and northeast Alabama and with two producer fields in Baldwin County. At the northeast site in 2019, CLRDV-infected PhytoGen 450 W3FE plants were shorter and produced fewer bolls and fruiting nodes, and there was an estimated seed yield loss of 5.8 percent. In central Alabama, accentuated verticality was noted on 14.3 percent of Deltapine 1646 B2XF plants, which tested positive for CLRDV, along with significantly higher counts of total and additional terminal nodes, but with fewer total bolls, fruiting nodes, and reduced seed yield of 5.1 percent. In the producer field, CLRDD- symptomatic NexGen 5007 B2XF plants displayed accentuated verticality, comprising 33.1 percent of the total stand, had greater terminal node counts compared with normal-appearing plants, which produced more bolls and fruiting nodes, along with 23 percent greater seed yield. Yield losses attributed to CLRDD were not observed in a second producer field in southwest Alabama.
In 2020, CLRDD did not impact yield in central Alabama; however, a significant yield reduction and reduced fruiting node counts were recorded for the CLRDV-positive compared with CLRDV- negative PhytoGen 500 W3FE plants at a southwest Alabama study site. Previously, a 2019 study estimated yield reduction of 560 kilogram/hectare (kg/ha) over 25 percent of Alabama’s 2018 cotton acreage, which translates into a statewide farm-gate income loss of $19 million. Compared with those in 2019, greatly reduced numbers of CLRDV-infected plants at several Alabama AAES units suggest that yield loss due to CLRDD in 2020 was low.
Management
As noted, disease incidence and subsequent symptom severity are much greater in late-planted cotton; therefore, in areas of Alabama at high risk for infection, cotton should be planted in early to mid-May. Early planting, which may increase the occurrence of boll rot and hardlock in south Alabama, has previously been recommended as a tool for managing CLRDV in cotton.
Long-term establishment of resistant cotton cultivars will be the best defense against poleroviruses like CLRDV and its relatives. Seed companies have germplasm with resistance to ‘typical’ and ‘atypical’ CLRDV strains, but these have proven susceptible to the US-CLRDV strain. Commercial cultivars available in the US are not immune to US-CLRDV, but observations indicate that cultivars do respond differently. Breeding trials are currently being conducted to evaluate a wide range of germplasm.
Henbit (Lamium amplexicaule), a common winter weed, white clover (Trifolium repens), evening primrose (Oenothera biennis), and other species have been found to be natural hosts of CLRDV and likely play a role in the overwintering of the virus and are a likely source of primary inoculum of US-CLRDV. Several species in the Malvaceae family have been reported as hosts for CLRDV under experimental conditions, including Cicer arietinum, Hibiscus sabdariffa and Sida acuta. The cotton aphid is a polyphagous species and is also reported to feed on plants in the Malvaceae family, but there is limited information on host-use patterns in the southeast. While not all the overwintering hosts in the US have been identified, an aggressive weed control program around field borders and nearby ditch banks may be helpful in delaying virus movement from overwintering hosts into cotton. In addition, producers are advised to destroy cotton stalks following harvest as well as kill ratoon and volunteer cotton, all of which are part of the green bridge that serves as a potential reservoir for overwintering virus and aphids.
Literature Cited
- Aboughanem-Sabanadzovic, N., Allen, T. W., Wilkerson, T. H., Conner, K. N., Sikora, E. J., Nichols, R. L., and Sabanadzovic, S. 2019. First report of Cotton leafroll dwarf virus in upland cotton (Gossypium hirsutum L.) in Mississippi. Plant Disease 103:1798.
- Aboughanem, N; Allen, T. W.; Wilkerson, T. H.; Scheffler, J.; and Sabanadzovic, S. 2021. Study of Cotton leafroll dwarf virus in Mississippi: State of the Art (2020). 2021 Beltwide Cotton Conference (in press).
- Agrofoglio, Y. C.; Delfosse, V. C.; Casse, M. F.; Hopp, H. E.; Kresic, I. B.; and Distefano, A. J. 2017. Identification of a new cotton disease caused by an atypical Cotton leafroll dwarf virus in Argentina. Phytopathology 107:369-376.
- Ali, A. and Mokhtari, S. 2020. First report of Cotton leafroll dwarf virus infecting cotton (Gossypium hirustum) in Kansas. Plant Disease 104:1880.
- Ali, A.; Mokhtari, S.; and Ferguson, C. 2020. First Report of Cotton leafroll dwarf virus from Cotton (Gossypium hirsutum) in Oklahoma. Plant Disease 104:2531.
- Alabi, O.J.; Isakeit, T.; Vaughn, R.; Stelly, D.; Conner, K.; Gaytan, B.; Villegas, C.; Hitzelberger, C.; De Santiago, L.; Monclova-Santana, C.; and Brown, J. 2019. First report of Cotton leafroll dwarf virus infecting upland cotton (Gossypium hirsutum L.) in Texas. Plant Disease 104:998.
- Avelar, S.; Schrimsher, D.W.; Lawrence, K.S.; and Brown, J.K. 2019a. First report of Cotton leafroll dwarf virus associated with cotton blue disease in Alabama. Plant Disease 103:592- 593.
- Avelar, S., Sobrinho, Roberto R.; Conner, Kassie; Nichols, Robert L.; Lawrence, Kathy S.; and Brown, Judith K. 2019b. Characterization of the complete genome and P0 protein for a previously unreported genotype of Cotton leafroll dwarf virus, an introduced polerovirus in the USA. Plant Disease 104:780-786.
- Brown, J. K. 2001a. Viral Diseases of Cotton. pp 48-49 In: Compendium of Cotton Diseases, Second Edition. Editors T. L. Kirkpatrick and C. S. Rothrock. American Phytopathological Society, St. Paul, MN.
- Brown, J. K. 2001b. Cotton Blue Disease. pp 50-51 In: Compendium of Cotton Diseases, Second Edition. Editors T. L. Kirkpatrick and C. S. Rothrock. American Phytopathological Society, St. Paul, MN.
- Correa, R. L.; Silva, T. F.; Simoes-Araujo, J. L., Barroso, P. A. V.; Vidal, M. S.; and Vaslin, M. F. S. 2005. Molecular characterization of a virus from the family Luteoviridae associated with cotton blue disease. Arch Virol 150:1357-1367.
- Distefano, A.; Bonacic Kresic, I.; and Hopp, H. 2010 The complete genome sequence of a virus associated with cotton blue disease, Cotton leafroll dwarf virus, confirms that it is a new member of the genus Polerovirus. Arch Virol 155:1849-1854.
- Faske, T. R.; Stainton D.; Aboughanem-Sabanadzovic, N.; and Allen, T. 2020. First report of Cotton leafroll dwarf virus from upland cotton (Gossypium hirsutum) in Arkansas. Plant Disease 104:2742.
- Hagan, A. K., Strayer-Scherer, A.; Burch, K.; Conner, K.; Bowen, K.; Miller, B.; Mclemoyl, C.; and Burkett, J. Assessment of yield loss attributed to Cotton leafroll dwarf disease in Alabama cotton. 2021 Beltwide Cotton Conference (in press).
- Iriarte, F.; Dey, K. K.; Small, I. M.; Conner, K.; O’Brien, K.; Johnson, L; Savery, C.; Carter, E.; Sprague, D.; Wright, D. L.; Nichols, R. L.; Mulvaney, M.; and Paret, M. L. 2020. First report of Cotton leafroll dwarf virus (CLRDV) in Florida. Plant Disease 104:2744.
- Michelotto, M. D. and Busoli, A. C. 2003. Effect of the time of inoculation of the Cotton vein mosaic virus by Aphis gossypii on growth and yield of cotton plants. Neotropical Entomology 35:251-256.
- Michelotto, M. D. and Busoli, A. C. 2009. Biology of cotton aphid on plants infected by Cotton vein mosaic virus. Bragantia 68:1017-1024.
- Mukherjee, A. K.; Chahande, P. R.; Meshram, M. K.; and Kranthi, K. R. 2012. First report of Polerovirus of the family Luteoviridae infecting cotton in India. New Disease Reports 25:22.
- Mukherjee, A. K.; Mukherjee, P. K.; and Kranthi, S. 2016. Genetic similarity between Cotton leafroll dwarf virus and Chickpea stunt disease associated virus in India. Pl. Path. J. 32:580- 583.
- Price, T.; Valverde, R.; Singh, R.; Davis, J.; Brown, S.; and Jones H. 2020. First report of Cotton leafroll dwarf virus in Louisiana. Plant Health Progress 21:142–143.
- Ray, J. D.; Sharman, M.; Quintao, V.; Rossel, B.; Westway J.; and Gambley, C. 2016. Cotton leafroll dwarf virus detected in Timor-Leste. Australian Plant Disease Notes 11:29.
- Sharman, M.; Lapbanjob, S.; Sebunruang, P.; Belot, J. L.; Galbieri, R.; Giband, M; and Suassuna, N. 2015. First report of Cotton leafroll dwarf virus in Thailand using a species-specific PCR validated with isolates from Brazil. Australian Plant Disease Notes 10:1-4.
- Silva, T. F.; Correa, R. L.; Castilho, Y.; Silvie, P.; Belot, J. L.; and Vaslin, M. F. 2008. Widespread distribution and a new recombinant species of Brazilian virus associated with cotton blue disease. Virol J 5:123.
- Silva, A. K. F.; Romanel, E.; Silva, T. F.; Castilhos, Y.; Schrago, C. G.; Belot, J. L.; and Vaslin, M. F. S. 2015. Complete genome sequences of two new virus isolates associated with cotton blue disease resistance breaking in Brazil. Arch Virol 160:1371-1374.
- Smith, R.; Graham, S.; Jacobson, A.; Strayer-Scherer, A.; Hagan, A. K.; Sikora, E.; Lawrence, K.; Brown, S.; and Li, S. 2021. Cotton Insect, Disease, Nematode, and Weed Control Recommendations for 2021. AL Coop. Ext. Sys. 2021 IPM-0415.
- Tabassum, A.; Bag, S.; Roberts, P.; Suassuna, N.; Chee, P.; Whitaker, J. R.; Conner, K. N.; Brown, J.; Nichols, R. L.; Kemerait, R. C. 2019. First report of Cotton leafroll dwarf virus infecting Cotton in Georgia, USA. Plant Disease 103:1803.
- Thiessen, L. D.; Schappe, T.; Zaccaron, M.; Conner, K.; Koebernick, J.; Jacobson, A.; and Huseth, A. 2020. First report of Cotton leafroll dwarf virus in cotton plants affected by cotton leafroll dwarf disease in North Carolina. Plant Disease 104:3275.
- Wang, H.; Greene, J.; Mueller, J. D.; Conner, K.; and Jacobson. A. 2020. First report of Cotton leafroll dwarf virus in cotton fields of South Carolina. Plant Disease 104:2532.
 Kassie Conner, Extension Specialist; Amanda Strayer-Scherer, Extension Specialist, Assistant Professor, Entomology & Plant Pathology; Austin Hagan, Emeritus Professor, Entomology & Plant Pathology; Jenny Koebernick, Assistant Professor, Crop, Soil & Environmental Sciences; Alana Jacobson, Associate Professor, Entomology & Plant Pathology; Kira Bowen, Alumni Professor, Entomology & Plant Pathology; Edward Sikora, Extension Specialist, Professor, Entomology & Plant Pathology; Scott Graham, Extension Specialist, Assistant Professor, Entomology & Plant Pathology; and Steve Brown, Extension Specialist, Assistant Professor, Crop, Soil & Environmental Sciences, all with Auburn University.
Kassie Conner, Extension Specialist; Amanda Strayer-Scherer, Extension Specialist, Assistant Professor, Entomology & Plant Pathology; Austin Hagan, Emeritus Professor, Entomology & Plant Pathology; Jenny Koebernick, Assistant Professor, Crop, Soil & Environmental Sciences; Alana Jacobson, Associate Professor, Entomology & Plant Pathology; Kira Bowen, Alumni Professor, Entomology & Plant Pathology; Edward Sikora, Extension Specialist, Professor, Entomology & Plant Pathology; Scott Graham, Extension Specialist, Assistant Professor, Entomology & Plant Pathology; and Steve Brown, Extension Specialist, Assistant Professor, Crop, Soil & Environmental Sciences, all with Auburn University.
New August 2021, Cotton Leafroll Dwarf Virus, ANR-2539











