Fish & Water

Algae concentrates, when used correctly, support rapid oyster larval growth, strong settlement, and the production of high‑quality oyster seed.
Eastern oyster hatcheries along the northern Gulf Coast produce both diploid and triploid oyster larvae for research and commercial aquaculture. Larvae are reared through the free-swimming stage of the life cycle. Oyster larval husbandry involves maintaining good water quality, often through regular water changes, and providing algae as a food source for growth and development. Algae can be sourced from cultured live algae or nonliving algae concentrate.
Cultured live algae species can be combined and modified to promote better larval growth and survival while supplementing oxygen through photosynthesis. However, live algae cultures require a large footprint, dedicated equipment, additional water use, and significant labor and energy costs to maintain.
Algal concentrates containing a similar mix of algal species have a small footprint, long shelf life, and eliminate the need for specialized labor to grow live algae. However, cells in algal concentrates can stick to surfaces, settle to the bottom of rearing tanks, and lack the benefit of oxygen production.
While the estimated cost per kilogram of algae is similar between live algae and algae concentrate, hatcheries limited with space and labor may benefit from using concentrates. Live algae production requires substantial space and labor, and there are a limited number of individuals with expertise in live algae culture for shellfish aquaculture.
Algae Concentrate Feeding

Figure 1. The oyster hatchery at the Auburn University Shellfish Laboratory.
Hatchery feeding rates of algae concentrate are based on larval eastern oyster size and density. To determine appropriate feed rates, reference the Algae Concentrate Feeding Calculator available on the Alabama Extension website at aces.edu.
To prepare for feeding, a dry, concentrated water conditioner (e.g., ChlorAm-X, AquaScience Technologies, LLC) is first mixed with approximately 500 milliliters of 3 percent sodium chloride (NaCl) solution at a rate of 0.12 grams of water conditioner per milliliter of algae concentrate to be used. The water conditioner is used to remove chlorine and chloramines from the algae concentrate mixture and prevent ammonia buildup in the larval culture tanks. The algae concentrate is measured into a graduated cylinder and then mixed with this solution.
The NaCl solution can be premixed in large batches by adding 30 grams of food grade, noniodized salt (Morton Culinox 999) per liter of dechlorinated tap water. This solution, when mixed with algae concentrate, aids in the dispersal of algae cells and prevents clumping. This mixture is poured through a 20-micron mesh bag and collected in a clean 5-gallon bucket to separate the algal cells further and filter out cells too large for larval consumption. Additional 1-micron filtered seawater is poured through the bag to rinse algae through the mesh to reach the desired feed volume.
Algae Concentrate Feeding Methods
Algae should be maintained at optimal density in larval tanks to ensure consistent access to food. This can be accomplished using a continuous feeding system or by feeding larvae in batches (two or three times daily).
Both methods have advantages and disadvantages. Batch feeding is reliable, requires no equipment, and is low maintenance, but personnel need to be available during batch feeding times. Continuous feeding provides a consistent food supply, but the pump system requires additional up-front costs and daily upkeep to prevent the buildup of algae and potential bacteria in the delivery lines. There is also the risk of system malfunction if it is not well maintained or if power is lost. With each feeding method, an initial amount of feed or the first batch feed should be added to the larval culture tanks immediately following a water change to bring the algae cell density to optimal levels, ensuring larvae can resume feeding when restocked.
These feeding methods may be used in combination. Ultimately, the method used depends on the resources available to the hatchery for personnel and equipment.
Batch Feeding
This method uses doses of algae concentrate mixture poured directly into larval culture tanks at two or more points throughout the day (figure 2). No further dilution is needed after the initial algae concentrate mixing and screening.

Figure 2. Container with prepared algae concentrate being poured into a larval tank for batch feeding.
Continuous Feeding
Continuous feeding involves a peristaltic pump system (figure 3) that systematically adds algae into the tanks, providing oyster larvae with consistent feed availability that can be adjusted throughout the larval cycle. Here, prepared algae concentrate is stored in a larger reservoir, continuously mixed with a small aquarium pump, and refrigerated. Peristaltic pumps (figure 4) then pull algae from the reservoir and pump it through silicone tubing (1/16 inch I.D.) to feed larvae at specified times. A programmable cycle timer controls the peristaltic pumps and feed timing. The algae concentrate mixture in the feed reservoir can be diluted to accommodate the pump dosage (e.g., 25 milliliters of algae mixture dosed for 2 minutes at 10-minute intervals = 3,000 milliliters per day).
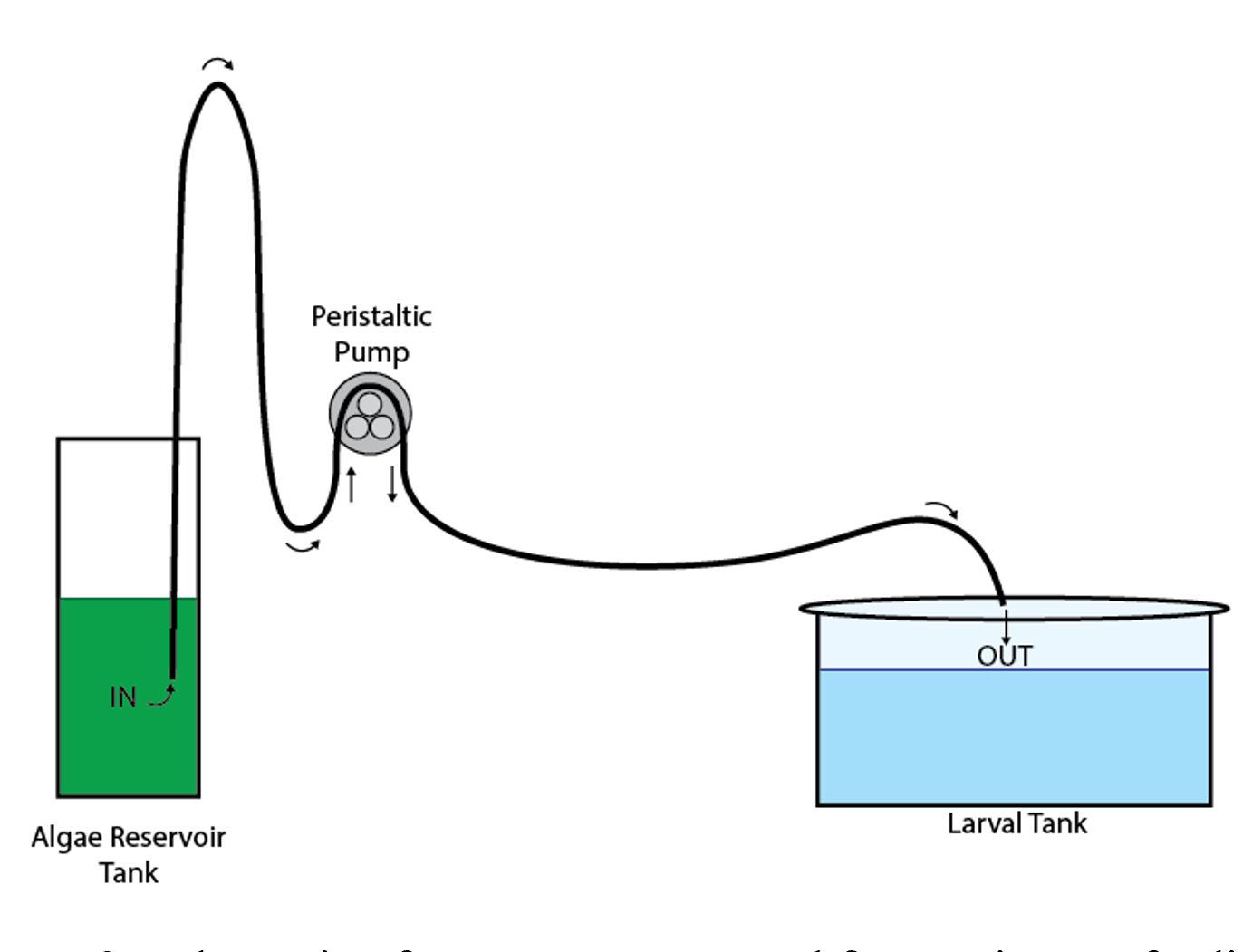
Figure 3. Schematic of pump system used in continuous feeding of algae concentrate.

Figure 4. Peristaltic pump system used in continuous feeding at Auburn University Shellfish Laboratory. Pumps (P) are connected to timers (T) that feed larvae throughout the day.
Continuous Feeding System Maintenance
Peristaltic pumps and silicone tubing that deliver feed to the tanks require daily cleaning. Before cleaning the tubing, the end that delivers algae to the tank (OUT in figure 3) should be removed from the culture tank and placed in a bucket to prevent contamination with algae sludge or bleach used during cleaning. Recommended cleaning procedures for the tubing consist of three steps: bleaching, stretching, and rinsing.
Bleaching. Make a 1 percent bleach solution by adding 10 milliliters of bleach to 1,000 milliliters of tap water and mixing thoroughly in a 1-liter beaker or cup. Place the IN end of the feed tubing into the bleach solution and initiate a 15-minute cycle of the peristaltic pumps to pump the bleach solution completely through the lines, emptying into the bucket.
Stretching. Stretching of the algae delivery lines should occur as the bleach solution runs through them. Beginning at the end of the tubing that pulls algae from the reservoir (IN), gently run wet fingers over the tubing with mild pressure to slightly stretch it and release algae sludge that may be clinging to the inside. Continue this process to the end of the tubing for each tank.
Rinsing. Replace the bleach water with a clean cup filled with tap water. Run the tap water through the tubing for 15 minutes to remove bleach residues. Finally, replace the tap water with 1-micron-filtered seawater and pump it through the tubing for 15 minutes to remove any remaining fresh water from the system.
The ends of the cleaned tubing can be reinserted into the feed reservoir and larval tanks. The prepared algae is ready for delivery.
Airlift Suspension of Algae Concentrates

Figure 5. Airlift setup for oyster larval tanks. The left image shows each system component, including a 4-inch PVC airlift pipe with a scalloped bottom edge, a 2-inch center PVC standpipe, and vinyl tubing with U-shaped component. The inset highlights the scalloped bottom edge of the airlift pipe. Yellow arrows indicate the locations of small holes for fitting the tubing. The right image shows the assembled airlift.
The beneficial use of algae concentrates can be enhanced by installing an airlift in the center of the tanks to keep the algae in suspension (figure 5). A 4-inch airlift PVC pipe is placed over the 2-inch center PVC standpipe. The end of the pipe closest to the tank bottom has small scallops cut along the bottom edge to allow water to enter the airlift and be driven toward the top of the tank. The top end of the airlift should be 4 to 6 inches below the water surface in the tank.
Two small holes are drilled in the bottom of the airlift and fitted with vinyl tubing to deliver pumped air to lift water in the airlift pipe. Open-ended air lines are used to create large bubbles that drive water up the airlift, resulting in a gentle, vertical, circular flow of water that enhances the suspension of the algae concentrate. Use of airstones is discouraged in tanks using algae concentrates, as the small bubbles they create can cause algae to clump.
Summary
Algae concentrates are an effective strategy for rearing oyster larvae along the northern Gulf Coast. Their use results in larval growth of approximately 15 to 20 µm
per day, and the average time to first set is 12 days postfertilization.
As concentrates may clump and fall out of suspension in larval tanks, care must be taken to prepare and distribute this algae. If applied appropriately, concentrated algae can be used to produce oyster larvae that grow quickly, set well, and produce high-quality seed.
 Andrea Tarnecki, Assistant Extension Professor, and Kayla Boyd, former Graduate Student, both in Fisheries, Aquaculture, and Aquatic Sciences, Auburn University
Andrea Tarnecki, Assistant Extension Professor, and Kayla Boyd, former Graduate Student, both in Fisheries, Aquaculture, and Aquatic Sciences, Auburn University
New April 2026, Algae Concentrates Used in Eastern Oyster Hatcheries, ANR-3247

