Private Well Program

Corrosivity is defined as the condition of water that causes it to dissolve metals, such as those in metallic plumbing, at an excessive rate. Learn how it can affect your plumbing system.
All water is somewhat corrosive, but these primary factors make water highly corrosive (sometimes called aggressive):
- Lower pH value (typically less than 7)
- Lower alkalinity (in this document, the term alkalinity will be used to specifically mean dissolved inorganic carbon)
- Higher specific conductivity, which is an indirect measure of “total dissolved solids”
- Higher temperature
Most surface water and many groundwater sources in Alabama and throughout the United States are highly corrosive. If you have copper plumbing and notice a bluish green stain on sinks and showers or below a leaky faucet, your water is highly corrosive. Blue green is the color of copper oxide, as orange brown is the color of iron oxide.
Effects of Corrosive Water
Corrosive water can cause physical damage to plumbing systems, create taste problems, and create a health risk. These three concerns are discussed below.
Damage to Plumbing
If your water is leaching metal from the plumbing of your home, eventually the plumbing will need repair or replacement. While all plumbing will be effected over time by the water it carries, corrosive water damages plumbing much more rapidly than less corrosive water.
Aesthetic Considerations (Taste and Appearance)
As higher levels of copper are dissolved into the water, the water develops a metallic taste (usually bitter). There is little or no taste from lead, even when the dissolved lead concentration is high enough to be a health risk. The presence of elevated copper can also stain clothing, water fixtures, and hair. Flushing the plumbing will reduce staining, but will not decrease the structural damage that corrosion does to metal plumbing.
Health Risk
Corrosive water, by itself, is not a health concern; orange juice, vinegar, and carbonated soft drinks are all considerably more corrosive than most surface and groundwater sources in Alabama and the United States. What is of concern, however, is that corrosive water can dissolve metals from the plumbing within your home and your well pump. Consuming excessive amounts of certain metals can present a health risk.
Pure lead plumbing, other metallic plumbing containing lead impurities, or copper plumbing with lead soldered joints may be present in many older homes (built prior to the 1950s). In addition, most of the copper pipes and fittings used in household plumbing prior to the mid- to late 1980s have sweated joints where lead solder was used. When corrosive water is present in such piping, and especially with low water flow, these metals can accumulate to concentrations that present a health risk to users if that stagnant water is regularly consumed.
This accumulation of metals is at its worst when the water sits for many hours (stagnant) in the plumbing system. This can occur late at night (11 p.m. to 6 a.m.) and during the day (9 a.m. to 4 p.m.), when family members are at school or work. Using stagnant water for drinking, for making coffee and tea, or for diluting juice concentrates can result in an excessive lead or copper intake. To avoid this health risk, flush the plumbing before collecting water for consumption.
Flushing to Reduce Copper and Lead
The purpose of flushing is to capture fresh water from your well or the municipal water main. Both sources are usually very low in lead or copper. Flushing is a process by which one runs cold water for a period of time to allow fresh water to enter the plumbing system. This flushed water can be used to water outdoor plants.
There are two methods to determine how long you need to flush the cold water tap:
Flush approximately 1 minute or until the water has turned cold (cold water indicates the arrival of fresh water from the well or municipal water main). Use the following chart to determine the approximate volume of water to flush to be assured that fresh water has arrived directly from the well or municipal water main.
Water Volume in 100 Linear Feet of Pipe
| Inside Diameter (Inches) | Gallons |
|---|---|
| 0.5 | 1 |
| 0.75 | 2.3 |
| 1 | 4.1 |
| 1.25 | 6.4 |
EPA Action Levels for Lead and Copper
EPA has established a special health-based category of drinking water standards for lead and copper since they enter water primarily as byproducts of corrosion. That category is called action level. The EPA’s action level for lead in a public water system is 0.015 milligrams per liter (mg/L); the action level for copper is 1.3 mg/L. The action level, which differs from the maximum contaminant level (MCL), has a special meaning that is used for all other health-based drinking water contaminants.
For corrosion testing in public water systems, at least nine out of every 10 samples must have lead and/or copper concentrations below the respective action level noted above. The samples must be collected in accordance with special sampling protocols, which includes some stagnation occurs. The action level is exceeded if lead or copper concentration is above the respective action level in more than 10 percent of these stagnant samples. If lead and/or copper concentration exceed the action level, public water utilities are required to take the following actions:
- A detailed study of the corrosive nature of the water source
- Initiation of appropriate corrosion prevention measures
- Immediate notification of system users about the failure to meet the action level(s)
For a private well serving just one home, meeting the action levels for lead and copper require that individual copper and lead test be taken under appropriate stagnant sampling conditions and be below the respective action level. Please note, however, that action levels only legally apply to public water systems.
Determining Water Corrosivity
 The best method for determining whether water is excessively corrosive is to follow EPA-recommended sampling protocol. The alternative is to infer the water’s corrosiveness from measured pH and alkalinity.
The best method for determining whether water is excessively corrosive is to follow EPA-recommended sampling protocol. The alternative is to infer the water’s corrosiveness from measured pH and alkalinity.
Determining Corrosivity by Copper and Lead Concentrations in Stagnant Samples
According to the Safe Drinking Water Act, the corrosiveness of water for public water systems is determined by taking samples for lead and copper collected in accordance with the following protocol.
- Collect 1 liter for each sample.
- Take samples under stagnant conditions (no flow for at least the previous 6 hours).
- Take samples at homes with lead soldered plumbing (built between 1982 and 1987).
- Take an appropriate number of samples based on the number of people served by that public water system.
If more than 10 percent of these samples have lead and copper above the action level, the water is considered too corrosive and certain actions are required. These actions often result in the addition of chemicals to neutralize the water’s corrosiveness.
Inferring Water’s Corrosiveness From pH and Alkalinity Data
The corrosive potential of water is determined primarily by the water’s pH and total alkalinity. Water pH is the most important single term for estimating corrosivity. By reviewing your existing water quality data, you can estimate your water’s corrosiveness by applying the following guidance.
- If the pH is below 6.0, the water is considered highly corrosive.
- If the pH is between 6.0 and 6.9, the water is somewhat corrosive, and stagnant testing is probably appropriate.
- If the pH is between 7.0 and 7.5, the water is probably notexcessively corrosive.
- If the pH is above 7.6, the water should not be particularly corrosive to metal plumbing.
It is a common misconception that water with a pH of 7 or slightly above is not corrosive. In fact, water with a pH of 7 can be some what corrosive if the water’s alkalinity is very low (below 30 mg/L). Many water sources, including some groundwater supplies, have a pH of less than 7.0 and have very low alkalinity (i.e. less than 30 mg/L). Natural rainfall is very corrosive to metal containers, because it has a pH of 5.6 or lower and essentially no alkalinity.
Testing for Corrosivity
Before taking a water sample for laboratory testing, you need to evaluate your plumbing. If your system has copper piping, you should test for copper and lead. If the system has galvanized iron pipe, you should test for cadmium, lead, and zinc (zinc does not create a health risk, but does cause taste problems). Cadmium and lead are common impurities in zinc used for galvanizing. If your plumbing is PVC plastic for both hot and cold water, you will be pleased to know that plastic pipe is unaffected by corrosive water and no further testing is necessary. If there is more than one type of piping in the house, you must determine what type of piping feeds the faucet at which drinking water is typically collected (usually the kitchen sink). You may experience some corrosion of faucets, even with PVC piping, but lead leaching from faucets alone is not a major problem.
In addition to plumbing, lead and other metals can come from your well pump or the piping connected to your home. Well pumps manufactured after January 1, 1996, have little lead. Leaching of lead from older submersible water pumps is discussed elsewhere. Water quality tests for metals from your home’s internal plumbing should be performed using two samples. The first-grab nonflushed sample should be collected from water that has been in the pipe for 6 or more hours—simulating the worst-case scenario for stagnant water periods. A second flushed sample should be collected after the water has been run long enough to remove all stagnant water. This sample will test the water supply for contamination.
The most logical procedure is to test the nonflushed condition first. If these samples test high for lead or copper, then flushed samples should be collected and tested to confirm that the source water is low in lead and copper. If the nonflushed samples test low for lead and copper, the water is noncorrosive and further testing is not needed.
Tests for copper, lead, and cadmium in Alabama should be conducted at a lab certified by EPA or the Alabama Department of Environmental Management (ADEM). A single test for lead or copper can cost $20 or more.
Reducing Corrosion Health Risk
If you are unsure of the corrosiveness of a water supply, use the following guidelines:
- Do not use hot water for cooking or coffee, draw cold water and heat it.
- Flush pipes before using water for drinking or cooking.
- Secure potable water from an outside source but make sure that source is safe and tested. (Bottled water is tested and widely available in supermarkets.)
Neutralizing Water Corrosiveness
Treatment to reduce corrosivity of drinking water often involves increasing some combination of the water’s pH and/or alkalinity. Widely used methods for doing this are discussed briefly below.
Low-Technology Approach
For dug wells where low cost is a significant part of the decision process, add a layer of calcite chips to the bottom of the existing well. Calcite is a form of limestone that is essentially pure calcium carbonate. The calcite will dissolve slowly, neutralizing the water’s corrosiveness. The thickness of calcite is determined by experimentation, but typically ranges from 3 to 5 inches. Advantages of this approach are low initial costs and no moving parts. The only disadvantage is that it adds some hardness to the water. Be sure to disinfect the well after the addition of calcite, and be certain that you do not damage or otherwise constrict water flow into your suction line or your pump. Replace lost (dissolved) calcite as needed, probably every 3 to 6 months.
For bedrock, driven point, and drilled wells, and where cost is not a significant factor, purchase an acid-neutralizing tank system. The unit must be plumbed into your water-distribution system. The tank is filled with calcite or another acid-neutralizing media and is the higher-tech version of adding calcite chips to a dug well. Other than greater initial costs for the neutralizing tank and plumbing to install it, the advantages and disadvantages are the same as above. Replace lost calcite or other neutralizing media as needed, probably every 3 to 6 months.
High-Technology Approach
A dilute solution of soda ash (sodium carbonate) or baking soda (sodium bicarbonate) is fed into the water stream on the discharge side of the well pump using chemical feed equipment. Both pH and total alkalinity of the water are raised to make the water less corrosive, but hardness of the water is not increased. Sodium hydroxide (lye) is a stronger neutralizing agent, but it is a very dangerous chemical and not generally recommended for homeowner use.
The choice of chemical feed equipment should be based on cost and the ability and willingness of the homeowner to maintain the system. For example, soda ash solution systems require routine inspection and filling of the chemical storage tanks every few weeks. The neutralizing tank system requires little or no maintenance other than replacing the calcite chips or other neutralizing media every 3 to 6 months.
Operating Acid Neutralizing Systems Calcite Systems
Calcite use requires little or no calibration and is easy to use. A pH of 8.3 is the maximum pH you can achieve by dissolving calcite in water. This is an ideal pH for corrosion protection, but pH values within the range of 7 to 7.5 are much easier to achieve. Where calcite treatment is used in a dug well, start with a 2- to 3-inch layer of calcite chips in the bottom of the well and add more calcite to get the desired pH.
Fill an acid-neutralizing tank system, which you can purchase or make yourself, to at least one-half capacity but not completely full. Purchased tank systems usually come already packed with calcite or other media. Recognize that the calcite will dissolve, and thus calcite will need to be supplemented periodically. The calcite tank should hold at least 1 cubic foot of calcite chips. Bigger or more tanks can be installed in parallel to improve neutralizing action without significantly reducing water flow.
Chemical Injection Units
Once installed, chemical injection type neutralizing systems need calibration to effectively neutralize corrosiveness and eliminate health risks. Every well has a different water quality profile, and thus the adjustment will vary from well t well. The typical adjustment approach proceeds as follows. Where bluish green staining has been present in the past, the neutralization process takes place in two steps. First, make a rough adjustment to the input of acid neutralizing media to reduce the majority of the water’s corrosiveness, using the disappearance of the bluish green staining s a guide to the amount of neutralizing chemical needed. Second, fine-tune the amount of chemical addition needed to optimize the neutralization, using laboratory testing if you want it precise.
A typical setup for chemical injection on a private well is shown in the following diagram.
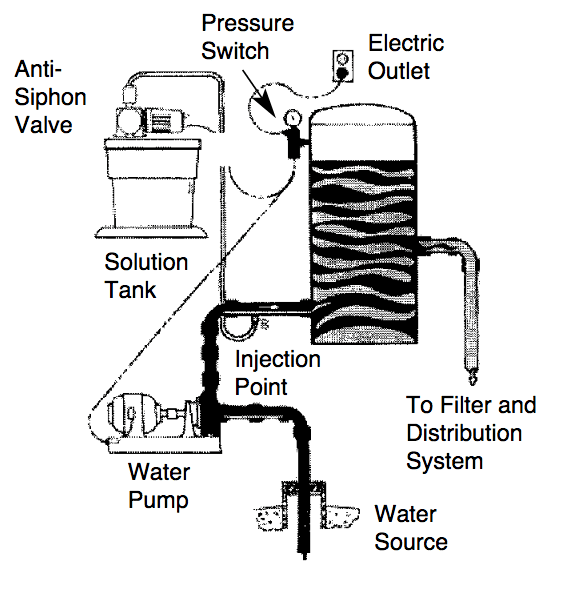
Typical setup for chemical injection on a private well.
The three steps for use and calibration of chemical injection units are given below.
- Clean any bluish green staining from a frequently used water fixture.
- Adjust the chemical feed level to that recommended by the company selling the device or, if no recommendation is made, select a relatively low amount of chemical. Allow a few weeks of operation for the bluish green staining to return. If the staining reoccurs, significantly increase the amount of neutralizing chemical, clean the stained water fixture, and observe for a few more weeks. Using the staining method to fine-tune avoids the expense of numerous laboratory testing.
- Once the bluish green staining is eliminated, or if staining was never a factor, the next step is to optimize the neutralization process. This requires laboratory testing for lead and copper in water quality samples collected after each subsequent small increase in chemical addition.
The samples should be collected under stagnant conditions, as discussed previously, and should be 1 liter in volume. Continue to increase chemical feed until both the lead and copper water quality test results are below their respective action levels. If the lead or copper levels remain high, contact a water quality professional for assistance.
James Hairston, former Extension Specialist, and Eve Brantley, Director of Water Resource Center, Crop, Soil, and Environmental Sciences, Auburn University
Reviewed October 2018, Water Corrosivity and Your Plumbing System, ANR-1230

