Crop Production

Bacterial spot is the most common disease of peppers in Alabama and is a recurring problem on tomatoes grown in the state. The disease can significantly affect marketable yield.
Bacterial spot is caused by four species of Xanthomonas (X. euvesicatoria, X. vesicatoria, X. perforans, and X. gardneri). These species were originally grouped together as the same species, Xanthomonas campestris pv. vesicatoria. Bacterial spot causes distinct symptoms on leaves and fruit and reduces both yield and fruit quality.
The main source of the pathogen is infected seed, infected transplants, infested crop debris, and contaminated equipment, such as stakes and tools. Fortunately, the bacterium cannot survive on its own in the soil. The pathogen can, however, survive on crop debris for up to a year, but typically this is not the case in the southeast where plant refuse breaks down rapidly. The pathogen can also survive on volunteer tomato and pepper plants in the field or in enclosed structures, such as greenhouses and high tunnels, during the winter.
Disease development is favored under moist, prolonged periods of high relative humidity (higher than 80 degrees F) and under temperatures between 68 degrees F and 95 degrees F. Night temperatures of 75 degrees F to 82 degrees F favor disease development while temperatures below 61 degrees F suppress disease progress regardless of daytime temperatures. Excess nitrogen levels favor the development of bacterial spot.
The pathogen can be active from the time of plant emergence through harvest. Infection is often initiated in the greenhouse during transplant production, with inoculum originating from infested seed. Warm, wet greenhouse conditions favor the spread of the pathogen to seedlings where it infects plants through wounds on leaves or stems such as those caused by insects or mechanical damage or through natural openings (i.e., hydathodes, stomates). Bacteria can colonize the transplants without any symptom development (latent infection); however, once planted in the field, bacterial spot symptoms start appearing. Thus, asymptomatic transplants harboring pathogen inoculum are often responsible for disease outbreaks in the field.
Symptoms of bacterial spot on tomato can appear on the leaves, fruit, and stems. Leaf lesions are dark and water soaked initially, eventually turning brownish black (figure 1). A narrow yellow halo may develop around the lesion that may resemble symptoms of the fungal disease early blight. However, early blight lesions develop a concentric ring pattern within their spots. Bacterial spot lesions appear sunken and greasy on the upper leaf surface with a translucent center and black margin. Spots are somewhat rounded but may be angular if restricted by leaf veins. The center of the spots frequently dries out and tears giving the leaf a ragged appearance (figure 2). Although initially about 1⁄8 inch in diameter, spots become poorly defined after a period of heavy moisture and coalesce, particularly along leaf margins. Only a few spots may cause a leaflet to turn yellow and drop prematurely. Lesions on the flower stems also cause the blossoms to die suddenly and drop.
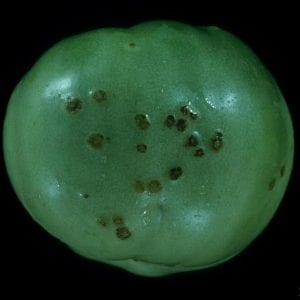
Spots on green tomato fruit first appear as small, black, raised pimples surrounded by a narrow water-soaked or greenish-white halo. Older spots are black, slightlyraised, and up to 1⁄3 inch in diameter (figure 3). In time, the raised centers develop into scabby, tan to brownish black sunken craters. Although spots remain small and do not penetrate deep into the fruit, large numbers will still reduce fruit quality. Ripe fruit appears to be resistant to infection by bacterial spot.
- Figure 1. Leaf lesions of bacterial spot can be brown to black. (Photo credit: David B. Langston, University of Georgia, Bugwood.org)
- Figure 2. The center of the spots frequently dries out and tears giving the leaf a ragged appearance.
- Figure 3. Older spots on green tomato fruit first appear as small, black raised pimples surrounded by a narrow water-soaked or greenish-white halo.
Symptoms of bacterial spot on peppers begin as small, yellowish-green, water-soaked spots on the lower leaf surface (figure 4). Spots first appear on older leaves in the lower canopy and spread upward with the aid of splashing water, high-pressure sprays, or the handling of wet plants by workers. These spots develop into small, circular, brown-to-black spots often surrounded by a narrow yellow halo. As the lesions expand, portions of the leaf or the entire leaf will turn yellow. Older spots increase in size up to 1⁄2-inch and turn tan to purplish-gray, often with dark brown margins (figure 5). The center of necrotic lesions often dries up and collapses, leaving holes in the leaf or ragged margins. Lesions eventually coalesce to form large, irregular dead areas on infected leaves. Severely infected leaves may wither and die at any time during the season. This leaf damage often causes significant defoliation of plants, especially in peppers, resulting in yield losses due to blossom drop and increased incidence of sunscald on fruit.
Spots on pepper fruit begin as dark, circular, water- soaked lesions up to 1⁄4-inch in diameter. Eventually, the spots become raised and cracked and have a warty appearance (figure 6). The spots are initially pale green but soon turn brown. During wet weather, various secondary decay-producing bacteria and fungi enter through these lesions, causing the fruit to rot before or after harvest.
- Figure 4. Symptoms of bacterial spot begin on the lower leaves. (Photo credit: Gerald Holmes, California Polytechnic State University at San Luis Obispo, Bugwood.org).
- Figure 5. Individual leaf spots on one leaf. (Photo credit: Paul Bachi, University of Kentucky Research and Education Center, Bugwood.org).
- Figure 6. Spots on pepper fruit eventually become raised and cracked and have a warty appearance. (Photo credit: Penn State Department of Plant Pathology & Environmental Microbiology Archives, Penn State University, Bugwood.org)
Management
Bacterial spot is extremely difficult to manage once established in a pepper or tomato field. This is why preventing the introduction of the pathogen on transplants is critical to managing this disease.
Management of bacterial spot requires an integrated pest management (IPM) approach employed from seed selection through harvest. For many years, management of the disease focused on the use of fixed copper sprays early in the season to suppress the bacterial population. Transplant houses would apply copper bactericides on 5- to 7-day intervals to keep the disease in check. Recommendations for fresh-market tomatoes consisted of initiating a copper-based spray program within one week after transplanting, with a minimum of three sprays on a weekly schedule. The copper product was often tank-mixed with a mancozeb-type fungicide to improve performance of the copper against the bacterium. This practice was effective in suppressing bacterial population early in the season in order to delay buildup of the disease. Emergence and rapid spread of copper resistance in the pathogen population, however, has rendered copper-based spray programs largely ineffective.
Host resistance has been a critical component of an IPM strategy to control bacterial spot on pepper. So far, eight resistance genes in pepper have been identified, and these have been incorporated into new cultivars using traditional breeding methods. Currently available commercial bacterial spot–resistant cultivars contain various combinations of these resistance genes.
Alabama Extension researchers have evaluated many of these cultivars as to performance and yield potential in Alabama. Currently, we recommend the bacterial spot resistant cultivars in table 1 for production in Alabama. All of the cultivars listed are hybrids. There are no open-pollinated cultivars with resistance to bacterial spot. When managing the disease, it might not be practical to identify which race(s) of the pathogen you are dealing with or which race(s) might become problematic. If bacterial spot has been a perennial issue, select a cultivar with resistance to all of the known races of this disease.
As observed in the past, durability of the host resistance depends on the changing pathogen population structure in response to the host selection pressure. Research surveys continue on the durability of current cultivars as changes occur in the pathogen population in Alabama fields. In tomato, four resistance genes have been identified to be effective against bacterial spot, of which Xv4 gene has been identified to be durable for the current pathogen population structure in the southeastern United States, and breeding efforts are currently underway. Currently, there are no commercial tomato cultivars available with resistance to bacterial leaf spot.
Bacterial spot and other bacterial diseases (i.e., bacterial speck, bacterial canker) multiply and spread rapidly under warm, wet conditions. For protectant bactericides to be effective, they must be applied prior to bacterial spot infection of the host. Because the immature fruit is most susceptible to infection, minimizing potential spread from the seed through the main fruit set will reduce potential losses due to the disease. It is also important to reduce plant stress (i.e., environment, soil compaction, poor fertility, pest pressure) by implementing a strategy that optimizes growing conditions to help reduce losses from bacterial spot.
Table 1. Recommended Tomato and Pepper Cultivars for Alabama
| Cultivar | Type of Pepper | Resistance to BLS RACE | Other Resistance |
|---|---|---|---|
| Alliance | Large-fruited (green to red) | 1 to 5 | PVY, CMV, TMV, PYMV, PMV |
| Aristotle | Large-fruited (green to red) | 1 to 3 | PVY |
| Camelot X3R | Large-fruited (green to red) | 1 to 3 | |
| Declaration | Large-fruited (green to red) | 1 to 5 | TSWV, CMV, Phytopthora root rot |
| Enterprise | Large-fruited (green to red) | 1 to 3 | |
| Excursion II | Large-fruited (green to red) | 1 to 3 | PVY, TMV |
| Green Machine | Large-fruited (green to red) | 0 to 10 | TSWV, TM |
| King Arthur | Large-fruited (green to red) | 2 | PVY, TEV, TMV |
| Mecate | Large-fruited (green to yellow) | 1 to 3 | PVY, TMV, PMV |
| Ninja S10 | Large-fruited (green to red) | 0 to 10 | TMV |
| Plato | Large-fruited (green to red) | 1 to 3 | TSWV, PVY |
| PS 09942815 | Large-fruited (green to red) | 1 to 10 | TSWV |
| Revolution | Large-fruited (green to red) | 1 to 5 | CMV, Phytopthora root rot |
| Samurai S10 | Large-fruited (green to red) | 0 to 10 | TMV |
| Sirius | Large-fruited (green to yellow) | 1 to 2 | TSWV |
| Vanguard | Large-fruited (green to red) | 1 to 5 | CMV, Phytopthora root rot |
| Wizard | Large-fruited (green to red) | 1 to 3 | PVY |
| Super Cayenne II | Cayenne | 2 | TMV |
| Compadre | Jalapeño | 2,5 | ToEV |
| El Rey | Jalapeño | 1 to 3 | |
| Ixtapa | Jalapeño | 1 to 3 | PVY |
| Tormenta | Jalapeño | 1 to 3 | PVY |
The first step in managing bacterial spot is selecting and purchasing the highest quality, disease-free certified seed from a reputable dealer. Ask for proof of seed certification before purchase. Seed can also be sanitized with hot water, sodium hypochlorite (bleach), or hydrochloric acid. Hot water seed treatments are the least effective of these options, but the easiest method to employ on-farm. Chlorox (bleach) seed treatment is the most effective of these strategies. One part Chlorox is mixed with 4 parts of water with 1 drop of surfactant. A pound of seed can be treated with 1 gallon of the above solution. Agitate the seed for 1 to 5 minutes, then rinse in cold water for 5 minutes and air dry seeds in a single layer.
Sanitation during transplant production is critical to managing this disease. Transplant producers need to remove all plant material from the greenhouse before starting a newcrop and between crops. Weeds and volunteer plants also need to be eliminated from in and around the production house. Growers should use sterile potting media and new trays, if possible. Do not reuse the seedling mix. If you are using compost, make sure it has been properly treated. All pots, flats, racks, tools, and equipment should be cleaned and disinfected before use. Disinfectants must be applied to clean surfaces to be effective. Wood can harbor the bacterium between seasons, so minimizing the use of wood in the greenhouse would be beneficial.

Figure 7. Avoid contact between seedlings from different seed lots as well as contact between tomato and pepper seedlings within the greenhouse to reduce potential cross contamination.
Avoid contact between seedlings from different seed lots as well as contact between tomato and pepper seedlings within the greenhouse to reduce potential cross contamination (figure 7). Minimize handling of seedlings and movement within the greenhouse. Minimize leaf wetness through proper timing of watering, and manage the relative humidity through adequate ventilation. Avoid high-pressure overhead watering, and water early in the day to allow time for foliage to dry. Avoid handling wet plants, and allow plants to dry before transferring them to a field. Scout the greenhouse regularly, and if symptoms of bacterial spot are observed, immediately remove the entire flat of plants as well as adjacent flats to prevent further spread of the pathogen. Dispose of these flats, and remove them from outside the greenhouse area as these could still act as a source of inoculum.
Bacteriophages are viruses that infect bacteria. Bacteriophages can be used in the greenhouse to reduce bacterial populations on the plant surfaces. These viruses are specific to the bacterial species present, and when in contact with bacterial cells, inject its RNA into the cell to cause the cell to break down. Treatments with these bacteriophages, marketed as AgriPhage, have been successful in reducing disease, especially in greenhouse transplant production. Efficacy of bactericides in managing the disease at transplant production has been fair to poor. Streptomycin-based products (i.e., Agri-mycin 17, Firewall, Streptol) can also be used during the transplant operation to manage bacterial populations on the surface of the transplants, with 4 to 5 applications typically needed beginning at the 2-leaf stage.
When picking up transplants from a distributor, don’t arrive with a trailer half loaded with plants from another greenhouse. Also clean and sanitize the trailer between loads. If holding plants overnight in a trailer, provide good ventilation to avoid a buildup of humidity and condensation in the trailer as this would promote development of the pathogen population.
Clean and sanitize the transplanter, focusing on surfaces that contact plants and trays, between fields, cultivars, or seed lots. Transplanting crews should clean and sanitize their hands at every break or change their disposable gloves. This prevents spread of the pathogen from one plant to another on workers’ hands.

Figure 8. Minimize leaf wetness through proper watering.
Try to rotate fields to a nonsolanaceous crop, such as squash, corn, or greens, for at least one year. Solanaceous weeds (i.e., black nightshade, ground cherry) can be the source of the bacterium and should be eliminated in the field before transplanting. Minimize stress to the crop by promoting good drainage and use a balanced fertility program. Use drip irrigation whenever possible and avoid the use of overhead irrigation to prevent water splashing and possible spread of the pathogen (figure 8).
Separate seed lots in the field in case one lot is infested with the disease. This will minimize spread within the field. Avoid planting tomatoes immediately adjacent to other host crops (i.e., peppers or earlier-planted tomato settings). Separate sequential plantings to avoid spread of the pathogen from an older setting to a younger set. For the same reason, work the youngest setting first during the day. Avoid performing crop maintenance operations, such as pruning, when the foliage is wet. Clean and sanitize tools for pruning and tying and have workers change disposable gloves at the end of each row to avoid spread of the bacterium. Eliminate hoeingand inter-row cultivating 3 to 4 weeks after transplantingto reduce leaf damage once rows begin to fill in.
Establish and maintain a balanced fertility program. Tomatoes and pepper crops growing under low fertility conditions are more susceptible to severe outbreaks of bacterial spot. Also avoid excessive fertility levels especially before fruit set as this can result in excessive foliage, low fruit set, and increased disease severity.
Application of fixed-copper products is still the primary method of managing bacterial spot in the field. Tank-mix the copper product with the fungicide mancozeb to increase the bactericidal effect of the copper. Actigard 50 WG (a.i. acibenzolar-S-methyl) is an SAR (systemic acquired resistance) product that can stimulate the plant’s own disease defense system. Actigard has been shown to reduce bacterial spot incidence and severity on tomatoes and should be used in combination with copper-based products in production fields. Always follow the manufacturer’s directions and restrictions when using any pesticide.
Plow crop debris soon after harvest is complete to help facilitate decomposition of the residue, eliminating the substrate on which the pathogen survives in the field. Burying the debris will also reduce the potential spread of the pathogen from an older setting that is out of production to later planted fields still producing fruit.
Edward Sikora, Extension Specialist, Professor, Entomology and Plant Pathology; Joseph Kemble, Extension Specialist, Professor, Horticulture; and Neha Potnis, Assistant Professor, Entomology and Plant Pathology, all with Auburn University.
Reviewed May 2022, Bacterial Spot of Tomato and Pepper, ANR-0819